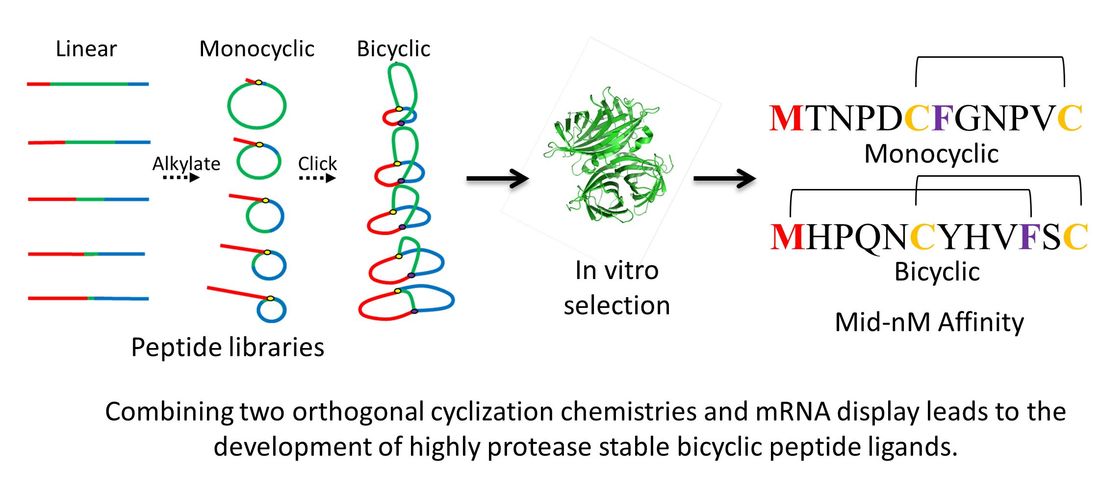
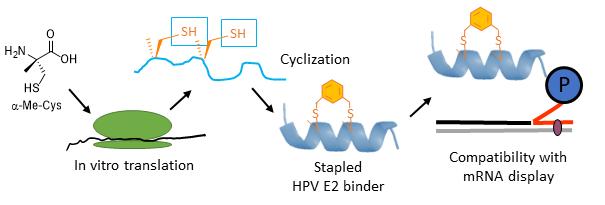
In Vitro Selections with Non-canonical amino acids and the synthesis of constrained peptide libraries
A common problem with peptides as potential therapeutics is that they are readily degraded by proteases. By replacing the 20 canonical amino acids in translation with modified "unnatural" variants, peptides can become protease resistant and more drug-like. We have shown that the ribosomal machinery used for protein synthesis can be hijacked and used to create peptides with unnatural building blocks. We have also used aminoacyl-tRNA synthetase engineering to expand the scope of unnatural building blocks usable in translation. These modified amino acids can then be used to create trillion-member libraries of peptides containing unnatural amino acids. These libraries, in turn, can be used to discover novel enzyme inhibitors or inhibitors of protein-protein interactions. In one application we have developed chemistry analogous to hydrocarbon stapling for the creation of alpha-helical peptide libraries. We have also developed a strategy for making highly constrained bicyclic libraries that are extremely resistant to degradation by proteases.